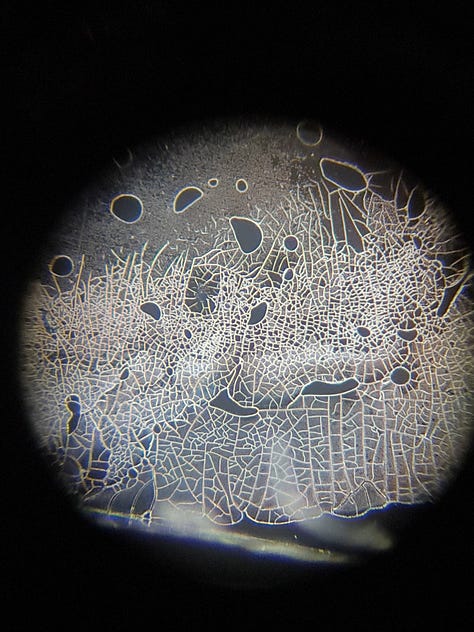
These videos show, in real-time, how quickly GQDs clump together under certain conditions (pH level - electrolytes, etc). (400 mag.)
Is this good for us, or not? Or, can aggregated GQDs do what they are supposed to do? Whatever that is...
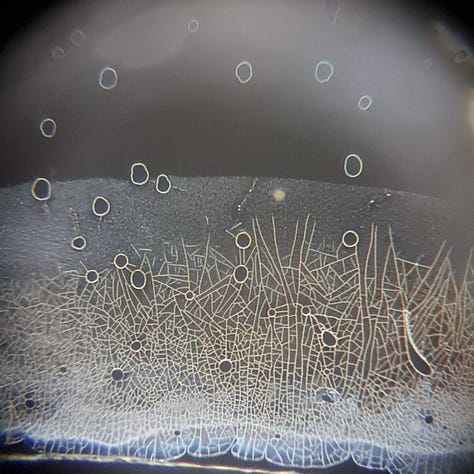
I checked it again about 2 hours later, and by then, it was gone:
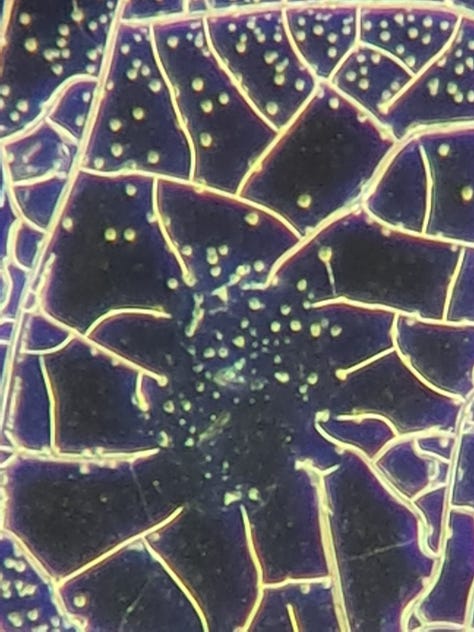
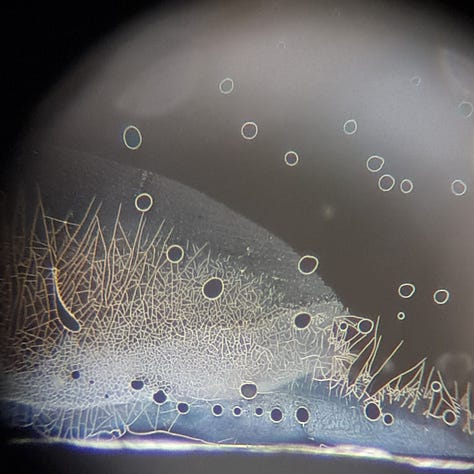
In the 2 years, I have never seen such a high level of aggregation, and I have never seen them disappear later. I only see aggregations every time, what remains on the slide. E.g.:
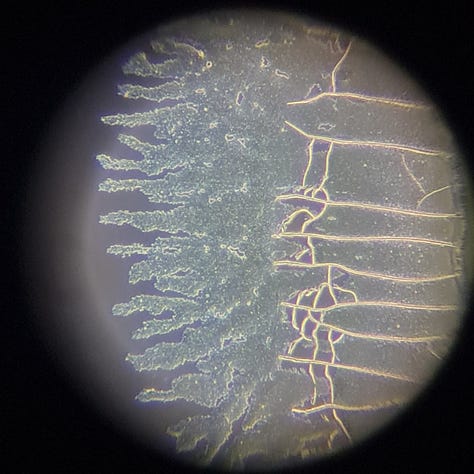
I think it's important to note that these aggregations are only visible at the edges of the slide. (Is it oxidation? Or something else?) There are only very few of these aggregated GQDs in the middle of the slide, usually on hydrogel-like formations or near them. For example:
Scipio gave me some research links to help me understand aggregation:
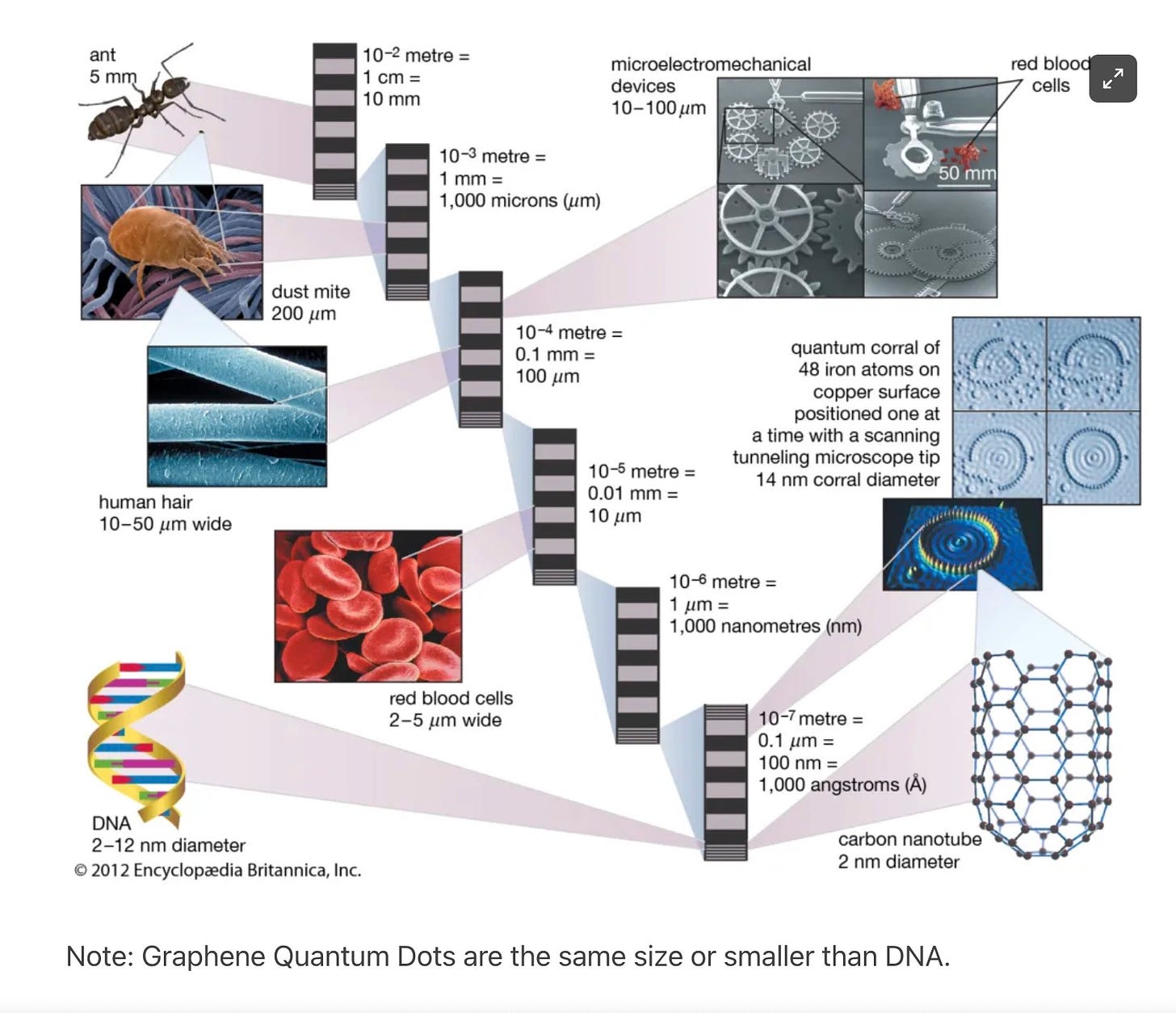
When quantum dots aggregate, they get so big that they can be seen with a microscope. A microscope with a magnification of 400x is enough to see aggregated quantum dots. (The size of a GQD is less than 100 nanometers, usually 2-8 nanometers.)
This size table comes from an article by Shawn Paul Melville:
As I mentioned above, I have been observing this phenomenon in my blood under a microscope for 2 years. I have not found any information about it on the internet so far.
The purpose of my posts is only to present the phenomena observed under the microscope and never to evaluate them, what they are or are not. (Since I am also a Data Analyst and sometimes a Product Manager (at the Art Department of film making), biology is not my field of expertise.)
For once, I will make an exception and quote from the above reference, which investigates the aggregation of GQDs in connection with changes in pH values (electrolytes).
I found this reference interesting because I have been drinking alkaline water (pH 9.2) for 2 years and taking electrolytes and supplements.
Just because I was drinking alkaline water didn't mean that the fluid between my cells was also alkaline. (Even though my diet was vegan, it did not consist of only alkaline foods.) This is proven by the pH value of my morning urine, which was around pH 6-pH 6.5. Since I drank about 2 liters of pH9.2 water during the day, the pH value of my urine during the day was around pH 8. I also had my blood tested during the day, so the blood’s pH was also alkaline. (What is good for my red blood cells :))
Here are some pictures about the ‘Art’:




Reference
The following data therefore come from the reference mentioned by Scipio:
Aggregation Kinetics and Self-Assembly Mechanisms of Graphene Quantum Dots in Aqueous Solutions: Cooperative Effects of pH and Electrolytes
I'm just copying a few quotes here. For a full understanding, please study the reference.
“…the response of GQDs to solutions with variable levels of electrolytes and pH could be of vital importance, as the GQDs may encounter concentrated cations, leading to variability in their transport and aggregation behavior.”
“The successive suppression of the electric double layer (EDL) leads to the gradual destabilization of nanoparticles, which is reflected by the lifting of attachment efficiency and the intensification of nanoparticle aggregation.”
“The colloidal behavior and aggregation mechanisms of GO have been studied previously. However, limited information concerning GQDs is available.’
“The overall objective of this work is to investigate the stability and aggregation kinetics of GQDs in aqueous systems with a wide range of water chemistries (i.e., cation type/valence, pH, and the combined effects of cations and pH). In this report, the cooperative effects of pH and electrolytes on the self-assembly and aggregation mechanisms of GQDs are unveiled and characterized.”
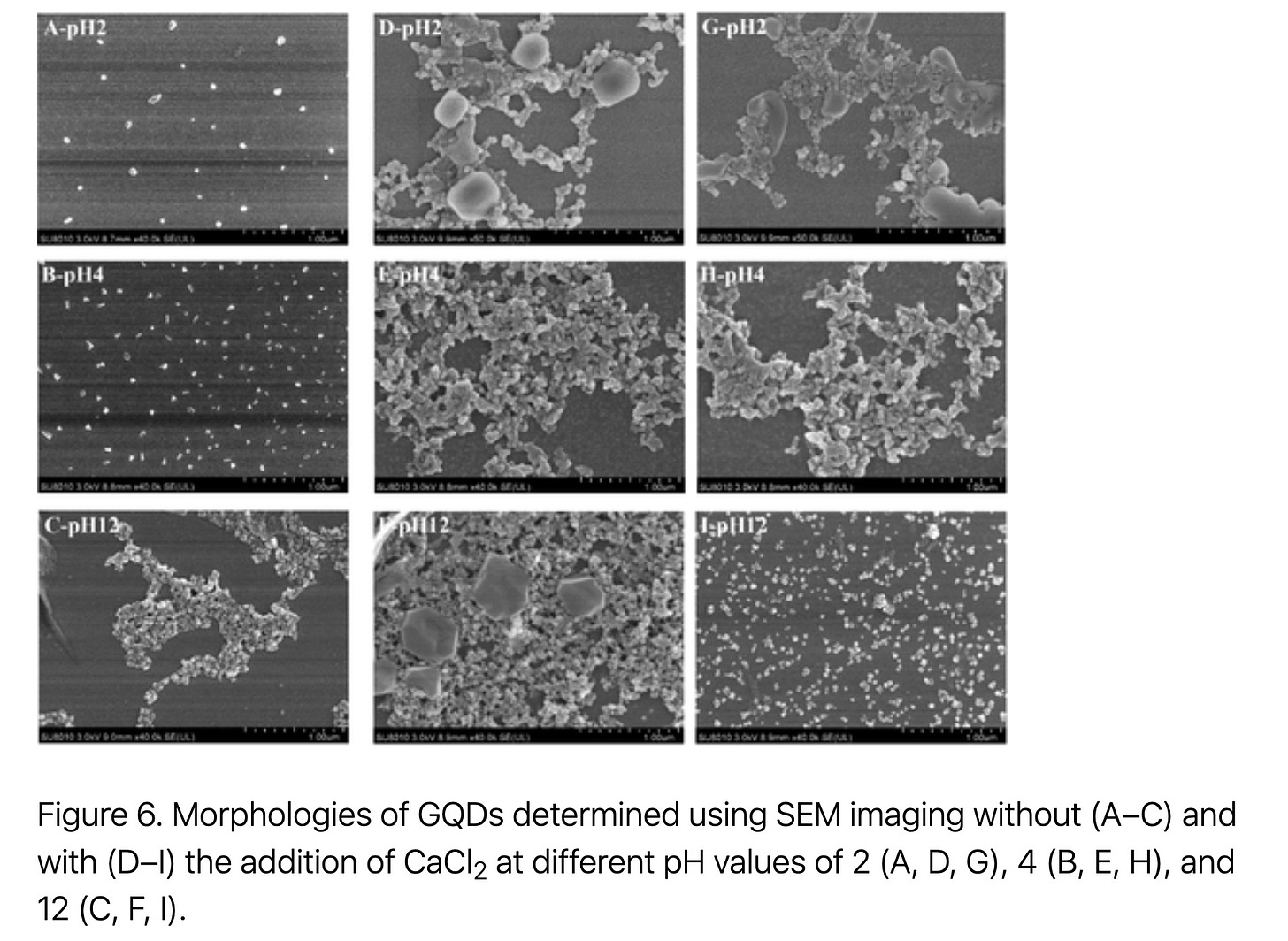
“The physical dimensions of the GQDs were between 3.9 and 26.8 nm (average square root of the area of GQD nanoplates, Table 1), with an average value of 9.0 nm according to the AFM particle analysis (Figure 1A).”
“To take a clear look at the effect of pH on the aggregates, the aggregation profiles of the GQDs at different pH values with the selected cations (at the CCC) were selected and are compared in Figure 5. All of the aggregation profiles are also available in Figure S-1 of the Supporting Information. In the presence of monovalent cations (Figure S-1A–F), the sizes of the aggregates were not significantly affected by pH. Only a slight decline in accordance with the pH increase was observed for NaCl (Figure 5A). This decline is clearly shown in Figure 5B for KCl. On the contrary, a large increase in aggregate size with pH elevation was observed in the case of the divalent cations (Figure S-1G–P). As is clearly shown in Figure 5C, the aggregate size at pH 12 was distinctly large. This is because Mg2+ primarily exists as a Mg(OH)2 colloid at pH 12, which enhanced the heteroaggregation of the GQD nanoplates and Mg(OH)2 colloids. Similarly, Chowdhury et al. recently studied the complex roles of divalent cations, pH, and NOM in the aggregation and stability of rGO. (35) An increase in the rGO aggregate size with pH elevation was observed in the presence of NaCl with CaCl2/MgCl2. The authors attributed this phenomenon to the stronger binding of Ca2+ with GO functional groups and the increased surface charge of GO.”

“The adjustment of the pH of the solution first changed the surface charge of the GQDs and made them act differently in acidic and alkaline solutions.”
“In contrast, when coexisting with divalent cations, GQD nanoparticles are heavily aggregated in solutions with a high pH (Figure 4C and D), which reveals a discrepancy with predictions based on the DLVO theory. Based on the proposed three-step aggregation mechanism, this colloidal discrepancy is attributed to the heavy self-assembly of GQDs (second step) induced by divalent cations, especially Ca2+, under alkaline conditions.”
“Our understanding of the assembly of GQD aggregates should be updated, as there could be aggregates that are stable in solutions, as well as aggregates that would precipitate out of solutions. These are significantly important findings related to the safety of GQDs as delivery vectors for therapeutics and the health risks of GQDs released into the environment.”
3 more videos:
This is my other fav. At 0:35, in the upper third of the image, a "donut" (aggregated GQDs) are sledding.



Incredible footage, you’ve captured the clearest video of Quantum Dot synthesis I’ve ever seen.
Crossposted this to my stack, keep up the good work!
Deadly art.